Publication Summary
Efficacy of an emollient cream in the treatment of xerosis in diabetic foot: a double-blind, randomized, vehicle controlled clinical trial
Objective
To evaluate the efficacy of an emollient cream (DEXERYL) on foot xerosis in diabetic patients.
Study
Prospective, randomized, multicenter, double-blind clinical trial. Contralateral vehicle-controlled study (placebo).
Population
57 diabetic patients.
Dosage
2 applications daily.
Duration
28 days.
Primary endpoint
To assess the severity of xerosis (XAS score) after 28 days of treatment with Dexeryl emollient cream compared with vehicle-treated feet.
Secondary endpoints
Change in XAS score at D14 and overall skin score. Number of feet with cracks, deep cracks, hyperkeratosis, threatening hyperkeratosis and xerosis at D14 and D28. Moisture index measured by corneometry and xerosis by image analysis on D-squame test at D14 and D28. Skin relief at D14 and D28. Subjects' opinions regarding treatment.
Tolerance
Dexeryl emollient cream is safe and well tolerated.
Conclusion
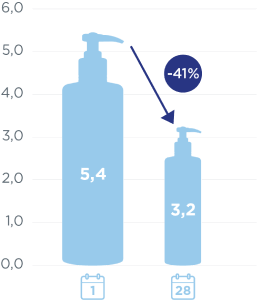
DEXERYL emollient cream significantly reduces foot dryness in diabetic patients.
Regular application of Dexeryl emollient cream twice a day to the feet for 28 days significantly reduces dryness in the feet of diabetic patients by 41%1. +61% increase in hydration index after 28 days1.
Publication
1 - MARTINI J. et al .Efficacy of an emollient cream in the treatment of xerosis in diabetic foot: a double-blind, randomized, vehicle controlled clinical trial. JEADV 2016
Want to read on?
This access is reserved for professionals, registered on Pierre Fabre For Med.
To access the full content, please register or log in if you already have an account.
