Summary of clinical results

Efficacy and safety of CLEANANCE COMEDOMED anti-blemishes concentrate - International observational study
Efficacy and tolerability of CLEANANCE COMEDOMED anti-blemishes concentrate prescribed alone, in combination with anti-acne drug treatments or as a relay, in adolescents and adults with mild to moderate acne
International observational study
Population
4230 subjects from 21 different countries, aged 12 to 25 and with mild to moderate facial acne (GEA score 1 to 3):
50.6% mixed acne (retentional and inflammatory)
36,2% retentional acne
13.2% inflammatory acne
Application of CLEANANCE COMEDOMED anti-blemishes concentrate
2 applications per day on the face for 2 to 3 months
- 46,9% as monotherapy
- 48.3% in combination with drug treatments
- 4.8% as relay treatments
Evaluation criteria
- Severity of acne (GEA score = Global Evaluation of Acne)
- Investigator-perceived efficacy investigators
- Quality of life questionnaire (CADI scale from 0 to 15)
- Skin tolerance
Results
- Highly significant improvement in acne severity score at follow-up visit (at 2-3 months): -36.4% (p<0.001, versus Inclusion)
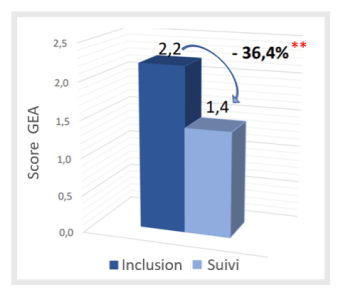
Change in acne severity score (GEA) after 2 to 3 months
** p= statistically Highly Significant (p<0.001, versus Inclusion)
- Investigator-perceived anti-infection effect in more than 80% of subjects in each population subgroup at follow-up visit (2-3 months)
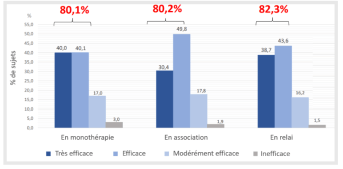
Investigator-perceived anti-acne efficacy in each subgroup after 2 to 3 months
- Highly significant improvement in quality-of-life score at follow-up visit (at 2 to 3 months): -44.2% (p<0.001, versus Inclusion)
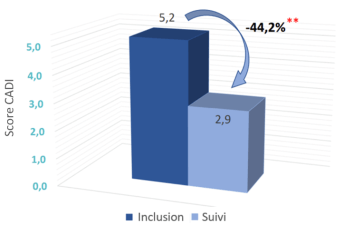
Evolution of quality of life score after 2 to 3 months
** p= statistically Highly Significant (p<0,001, versus Inclusion)
- Good to very good skin tolerance for 94.7% of subjects
Conclusion
CLEANANCE COMEDOMED anti-blemishes concentrate applied twice daily demonstrated anti-acne efficacy and quality of life benefit in a large international cohort
Good to very good skin tolerance
More summaries of clinical results
Want to read on?
This access is reserved for professionals, registered on Pierre Fabre For Med.
To access the full content, please register or log in if you already have an account.